11th Class CBSE Board Chemistry Mid Term Examination (2025-26)
St. Margaret Sr. Sec. School, Prashant Vihar, Sector 14, Rohini, Delhi-85, India
| School Name: | St. Margaret Sr. Sec. School, D-BLOCK, Prashant Vihar, Sector 14, Rohini, Delhi-110085 |
| Class: | 11th Standard (CBSE) |
| Subject: | Chemistry |
| Time Duration: | 03 Hours |
| Maximum Marks: | 70 |
| Date: | 28 / 12 / 2025 |
General Instructions: 11th Class Chemistry Exam 2025-26
This question paper contains 16 questions.
- The paper is divided into 5 sections: A, B, C, D and E.
- Section A consists of Multiple Choice Questions.
- Section B consists of Very Short Answer Questions.
- Section C consists of Short Answer Questions.
- Section D consists of Long Answer Questions.
- Section E consists of Numerical / Reasoning Questions.
- In the case of MCQs, the text of the correct answer should also be written.
SECTION – A
Question 01: The IUPAC name of the following molecule is:
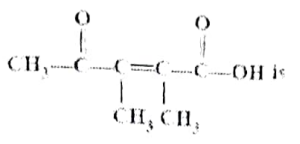
a) 4-oxo-2,3-dimethyl pent-2-en-1-oic acid
b) 2-carboxy-3-methyl pent-2-en-3-one
c) 4-carboxy-3-methyl pent-3-en-2-one
d) 2,3-Dimethyl-4-oxo-pent-2-en-1-oic acid
Question 02: Ionic species are stabilised by dispersal of charge. Which of the following carboxylate ion is the most stable?
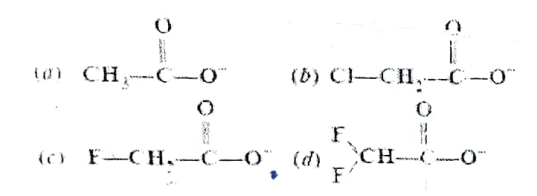
a) Option (a)
b) Option (b)
c) Option (c)
d) Option (d)
Question 03: Hyperconjugation involves delocalisation of ______.
a) electrons of C–H bond of an alkyl group directly attached to an atom of an unsaturated system
b) electrons of C–H bond of alkyl group directly attached to a positively charged carbon atom
c) π-electrons of carbon–carbon bond
d) lone pair of electrons
Question 04: The IUPAC name of the following compound is:
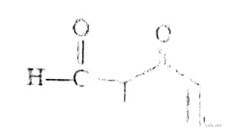
a) 5-Formyl hex-2-en-3-one
b) 5-methyl-4-oxohex-2-en-5-al
c) 3-keto-2-methyl hex-5-enal
d) 3-keto-2-methyl hex-4-enal
Question 05: The compound formed in the positive test for nitrogen in Lassaigne’s extract is:
a) Fe₄[Fe(CN)₆]₃
b) Na₃[Fe(CN)₆]
c) Fe(CN)₃
d) Na₄[Fe(CN)₅NOS]
Question 06: In Dumas method, 0.25 g of an organic compound gave 40 mL of N₂ at 300 K and 725 mm pressure. If aqueous tension at 300 K is 25 mm, the percentage of nitrogen is:
a) 16.75%
b) 15.76%
c) 17.36%
d) 18.20%
Question 07: Homolytic fission leads to formation of:
a) Nucleophile
b) Carbanion
c) Free radical
d) Carbocation
Question 08: In which of the following compounds the carbon marked with asterisk is expected to have greatest positive charge?
a) *CH₃–CH₂–Cl
b) *CH₃–CH₂–Mg⁺Cl⁻
c) *CH₃–CH₂–Br
d) *CH₃–CH₂–CH₃
Question 09: The correct representation involving heterolytic fission of CH₃–Br is:

a) Option (a)
b) Option (b)
c) Option (c)
d) Option (d)
Question 10: In the given organic compound, the pair of hybridised orbitals involved in the C₂–C₃ bond is:
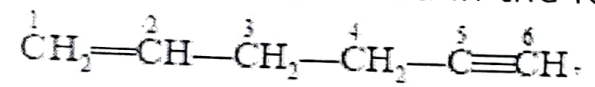
(i) sp – sp² (ii) sp – sp³ (iii) sp² – sp³ (iv) sp³ – sp³
SECTION – B
Question 11: Answer the following:
a) What type of isomerism is shown by:
i) Ethanal and Dimethyl ether
ii) Diethyl ether and Methyl propyl ether
b) Why does AlCl₃ act as an electrophile?
Question 12: Identify the most stable species in the following sets of ions, giving reasons:
i) CH₃· , CH₃Br· , CHBr₂· , CBr₃·
ii) CH₃· , CH₃Cl· , CHCl₂· , CCl₃·
SECTION – C
Question 13: Draw the resonance structures for the following compounds. Show electron shift using curved-arrow notation:
i) Aniline
ii) C₆H₅–NO₂
Question 14: Explain the terms inductive and electromeric effects. Which electron-displacement effect explains the acidity order:
Cl₃CCOOH > Cl₂CHCOOH > ClCH₂COOH
SECTION – D
Question 15: In Kjeldahl’s method, 0.50 g of an organic compound released ammonia which was absorbed in 50 cm³ of 0.5 M H₂SO₄. The residual acid required 60 cm³ of 0.5 M NaOH. Find the percentage of nitrogen.
Question 16: Answer the following:
a) Write IUPAC names of the given compounds 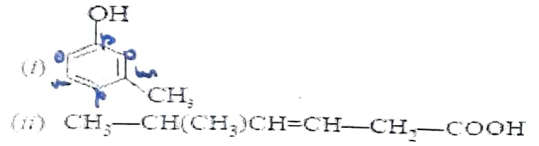
b) Which is more stable: O₂N–CH₂CH₂O⁻ or CH₃CH₂O⁻ ? Give reason.
 Class Notes NCERT Solutions for CBSE Students
Class Notes NCERT Solutions for CBSE Students





