10 CBSE Science Paper Design (2012-13) Outside Delhi (Modified Based on Latest CBSE Question Paper Design)
Class: 10th Class CBSE
Subject: Science
Time Allowed: 3 Hours
Maximum Marks: 80
General Instructions:
- The Question paper comprises of two sections, A and B. You are to attempt both the sections.
- All questions compulsory.
- All questions of Section A and all questions of Section B are to be attempted separately.
- Question number 1 to 2 in Section A are one mark questions. These are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section A are two marks questions. These are to be answered in about 30 words each.
- Question numbers 6 to 15 in Section A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section A are five marks questions. These are to be answered in about 70 words each.
- Question numbers 22 to 27 in Section B are questions based on practical skills and are two marks question.
Section A
Question: 1. Name the physical quantities which are indicated by the direction of thumb and forefinger in the fleming’s right hand rule? [1]
Answer: In Fleming’s right hand rule, forefinger points in the direction of magnetic field and thumb points in the direction of motion of conductor.
Question: 2. State modern periodic law of classification of elements. [1]
Answer: Modern periodic law states “that the properties of elements are a periodic function of their atomic numbers”.
Question: 3. Identify the type of reaction from the following equations: [2]
- CH4 + 2O2 + 2H2O
- Pb (NO3)2 + 2KI → PbI2 + 2KNO3
- CaO + H2O → Ca(OH)2
- CuSO4 + Zn → ZnSO4 + Cu
Answer:

Question: 4. State the importance of chromosomal difference between sperms and eggs of humans. [2]
Answer: A male has one X chromosome and one Y chromosome. Thus half the sperms will have X chromosomes and the other half will have Y chromosome.
A female has two X chromosomes. So all the female gametes will have only X chromosomes. If a sperm carrying Y chromosome fertilizes an ovum then the child born will be a boy.
Thus the chromosomal difference between sperms and eggs of humans determines the sex of the child.
Question: 5. “A concave mirror of focal length ‘ f ‘ can form a magnified erect as well as an inverted image of an object placed in front of it.” Justify this statement starting the position of the object with respect to the mirror in each case for obtaining these images. [2]
Answer: When the object is placed at a distance less than ‘f ‘ (focal length) from the mirror, the image formed is virtual, magnified and erect.
When the object is placed at a distance of ‘f ‘ (focal length) from the mirror, the image is real, inverted and highly magnified.
When the object is placed at a distance more than ‘f ‘ (focal length) but less than ‘2f ‘ , the image formed is real, inverted and magnified.
When the object is placed at the distance of ‘2f ‘ from mirror, the image is real, inverted and same size of the object.
When the object is placed is placed at a distance more than ‘2f ‘ from the mirror, the image formed is real, inverted and diminished.
Question: 6.
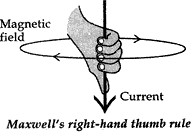
- State Maxwell’s right-hand thumb rule.
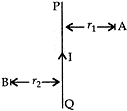
- PQ is a current carrying conductor in the plane of the paper as shown in the figure. Mention the direction of magnetic fields produced by it at points A and B. Given r1 < r2, where will the strength of the magnetic field be larger? Justify your answer in each case. [3]
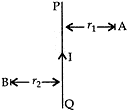
Answer: (i) Maxwell’s right hand thumb rule. The direction of the current is given by Maxwell’s right hand thumb rule, “If the current carrying conductor is gripped with the right hand in such a way that the thumb gives the direction of the magnetic field produced around the conductor”.
(ii). Since the direction of current in the straight conductor is from Q to P, then according to Maxwell’s right hand thumb rule, magnetic field at point A is inside the paper and at point B is outside the paper. Since r1 < r2 the strength of the magnetic field at A is more than at B because greater the distance of a point from the current carrying wire, weaker will be the magnetic field produced at that point.
Question: 7. 2g of ferrous sulphate crystals are heated in a boiling tube.
- State the colour of ferrous sulphate crystals both before heating and after heating.
- Name the gases produced during heating.
- Write the chemical equation for the reaction. [3]
Answer: (i) The colour of the ferrous sulphate crystals is green before heating and changes to brown after heating. The colour of the crystals changes due to the formation of ferric oxide.
(ii) During heating sulphur dioxide (SO2) and sulphur trioxide (SO3) are produced.
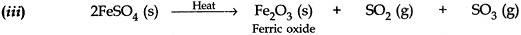
Question: 8. Give reason for the following:
- Electric bulbs are usually filled with chemically inactive gases like nitrogen and argon.
- Copper and aluminium wires are usually employed for electricity transmission.
- Fuse wire is placed in series with the device. [3]
Answer:
- Electric bulbs are usually filled with chemically inactive gases like nitrogen and argon because these gases do not react with the hot tungsten filament and hence prolong the life of filament of the electric bulb.
- Copper and aluminium wires are usually employed for electricity transmission because copper and aluminium have very low resistivity and thus they are very good conductors of electricity.
- Fuse wire is placed in series with the device because when large current passes through the circuit the fuse wire gets heated up and melts and whole circuit breaks and the device is protected from the damage.
Or
(a) Shruti draws magnetic field lines close to the axis of a current carrying circular loop. As she moves away from the center of circular loop, she observes that the lines keep on diverging. Explain the reason for her observation.
(b) Write two properties of magnetic field lines.
Answer: (a) The pattern of magnetic field due to current carrying circular loop are circular near the current carrying loop but the concentric circles representing magnetic field lines become bigger and bigger. At the centre of the circular loop, the magnetic field lines are straight. Therefore on moving away from the centre of circular loop, magnetic field lines keep on diverging.
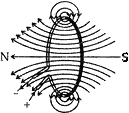
(b) Properties of magnetic field lines:
(i) These lines originate from the north pole and end at the south pole.
(ii) Two magnetic field lines do not intersect each other.
Question: 9. Name three different glands associated with the digestive system in humans. Also name their secretions. [3]
Answer: Three glands associated with the digestive system are as follows:
- Salivary glands in the mouth produce saliva. Saliva contains an enzyme called salivary amylase which digests the starch present in food into sugar.
- Liver is the largest gland which secreted bile and pours its secretion in the duodenum (part of the small intestine). Bile makes the acidic food coming from the stomach alkaline so that pancreatic enzymes can act on it. Bile salts also break the fats present in the food into small globules.
- Pancreas is also a large gland which secretes pancreatic juice into the duodenum. Pancreatic juice contains
(i) pancreatic amylase which breaks down the starch.
(ii) trypsin digests the protein.
(iii) lipase which breaks down the emulsified fats.
Pancreatic juice acts on alkaline medium.
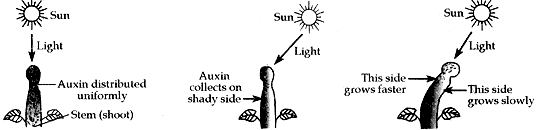
Question: 10.
- Name the hormone which is secreted when growing plants detect light. Mention its site of secretion in a plant.
- Explain why do plants appear to bend towards light?
Answer:
- Auxin hormone is secreted in the growing plant which detects light.
(i) It is present in the tip of the growing stem. - Plants appear to bend towards light. As more auxin is produced on the shaded side than on the lighted side of the stem, so stem grows faster on the shaded side than on the lighted side.
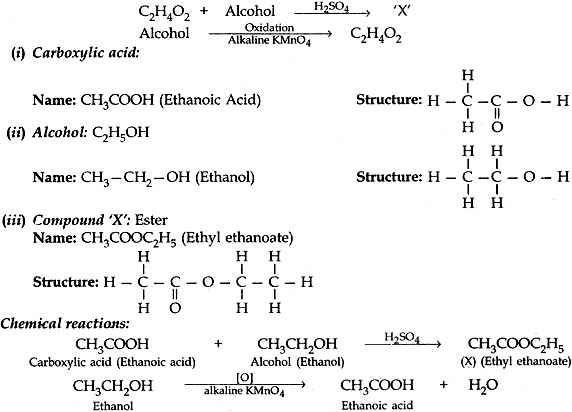
Question: 11. A carboxylic acid C2H2O2 reacts with an alcohol in the presence of H2SO4 to from a compound ‘X’. The alcohol on oxidation with alkaline KMnO4 gives the same corbonxylic acid, C2H4O2. Write the name and structure of (i) Carboxylic acid, (ii), alcohol and (iii) the compound ‘X’. [3]
Answer: c, C2H2O2:
Question: 12. The electronic configuration of an element ‘X’ belong? State its valency. Justify your answer in each case. [3]
Answer: Electronic configuration of ‘X’ is 2, 8, 8, 2.
- It belongs to the 4th period because it has four energy shells and period number of an element is equal to the number of energy shells.
- It belongs to 2nd group because it has 2 valence electrons and the group number of an element having upto two valency is ‘2’. To acquire inert gas configuration ‘X’ either loses 2 electrons or gains 6 electrons. Since the shorter route is to lose 2 electrons for obtaining inert gas configuration, hence its valency is 2.
Or
Four elements P, Q, R and S have atomic numbers 12, 13, 14 and 15 respectively. Answer the following questions giving reasons:
- What is the valency of Q?
- Classify these elements as metals and non-metals.
- Which of these elements will form the most basic oxide?
Answer:
| Elements | Atomic No. | Electronic configuration | Valence electrons |
| P Q R S |
12 13 14 15 |
2, 8, 2 2, 8, 3 2, 8, 4 2, 8, 5 |
2 3 4 5 |
- Valency of Q is 3 as Q loses 3 electrons to obtain inert gas configuration.
- P and Q are metals as these elements lose 2 and 3 elements respectively to obtain inert gas configuration and those elements which lose electrons are metals.
R and S are non-metals as these elements either share or gain electron for attaining inert gas configuration and those metals which accept electrons either by sharing or gaining are non-metals. - Element P Will from the most basis oxide because P is the most metallic element among the 4 given elements and thus forms the most basic oxide.
Question: 13. Explain with the help of suitable example why certain traits cannot be passed on to the next generation? What are such traits called? [3]
Answer: A trait (or characteristic) of an organism which is ‘not inherited’ but develops in response to the environment is called and acquired trait.
The acquired traits of an organism cannot be passed on to its future generations.For example, ‘low weight’ of beetle, ‘cut tail’ of a mouse.
Changes brought in the non-reproductive tissues cannot be passed on to the DNA of the germ cells. So the experiences of an individual during its lifetime cannot be passed on to its progeny. Thus acquired characters of an individual cannot be inherited.
Question: 14. Name the type of mirror used (i) by dentists and (ii) in solar furnaces. Give two reasons why such mirrors are used in each case. [3]
Answer:
- Concave mirrors are used by dentists to see the large images of the teeth of patients because when a tooth is within the focus of a concave mirror, then an enlarged image of the tooth is seen in the concave mirror. Thus it becomes easier to locate the defect in the tooth.
- Large concave mirrors are used in solar furnaces as reflectors. Solar furnace is placed at the focus of the concave reflector which focuses the Sun’s heat rays on the furnace due to which the solar furnace gets very hot Even steel can be melted in this solar furnace.
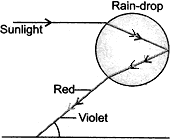
Question: 15. When and which do we see a rainbow? How is a rainbow formed? Draw a labelled diagram to illustrate the formation of a rainbow. [3]
Answer: Rainbow is caused by dispersion of sunlight by tiny water droplets present in the atmosphere. The water droplets act like small prisms. They reflect the incident sunlight and then reflect it internally and finally refract it again when it comes out of the rain-drop. A rainbow is always formed in the direction opposite to that of the Sun.
Question: 16.
- Explain the steps for extraction of copper from its ore. Write the reaction involved.
- Draw a neat, labelled diagram for electrolytic refining of copper and explain the process. [5]
Answer: (a) Copper is found as Cu2S in nature. The concentrated Cu2S is roasted in presence of air and Cu2O is formed.
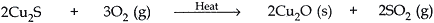
Copper oxide (Cu2O) is then heated in the absence of air. This reduces Cu2O into copper metal. The copper obtained is called blister copper because the SO2 produced during this reaction gets trapped inside its surface causing blisters to appear on the surface of copper metal.

Or
- Show the formation of magnesium chloride and sodium chloride by transfer of electrons.
- Identify the ions present in these compounds.
- Why do ionic compounds not conduct electricity in the solid state?
Answer: (a) (i) Sodium (Na) and Magnesium (Mg) have 1 and 2 electrons respectively in the outermost shells of their atoms, so they lose their electrons to achieve the inert gas electron configuration of eight valence electrons and form positively charged ions or cations.

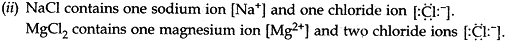
(iii) Ionic compounds in the solid state do not conduct electricity because movement of ions in the solid state is not possible due to their rigid structure.
Question: 17.
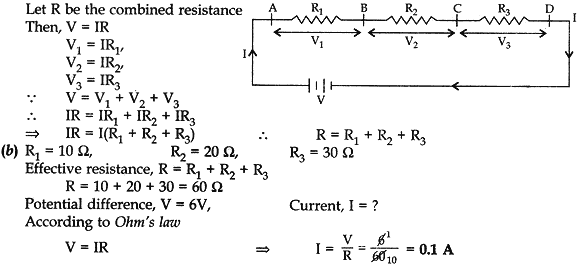
- Resistors given as R1, R2, and R3 are connected in series to a battery V. Draw the circuit diagram showing the arrangement. Derive an expression for the equivalent resistance of the combinations.
- If R1 = 10 Ω, R2 = 20 Ω and R3 = 30 Ω, calculated the effective resistance when they are connected in series to a battery of 6 V. Also find the current flowing in the circuit. [5]
Answer: (a) Same current (I) flows through different resistances, when these are joined in series, as shown in the figure:
Question: 18. (a) Draw a diagram of human alimentary canal and label the following parts:
- Largest gland.
- gland that secretes digestive enzymes and hormone.
- part where HCl is produced.
- part where digested food is absorbed.
What are villi? Explain their function in the digestive system. [5]
Answer: (a) (i) Largest gland – Liver
(ii) Gland that secretes digestive enzymes and hormone – Pancreas
(iii) Part where HCl is produced – Small intestine
(iv) Part where digested food is absorbed – Stomach
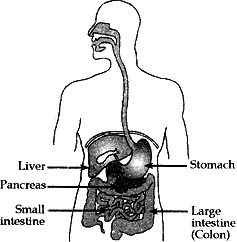
(b) The small intestine is especially adapted for absorption of digested food. The inner surface of small intestine has millions of tiny, finger like projections called villi which provide the inner walls of small intestine a very large surface area. The large surface area helps in the rapid absorption of digested food. The villi are richly supplied with blood vessels which take the absorbed food to each and every cell of the body.
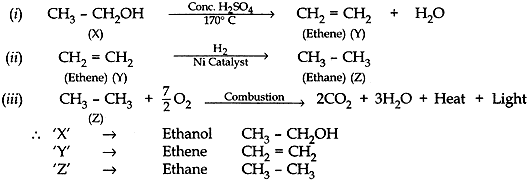
Question: 19. An organic compound ‘X’ on heating with conc. H2SO4 forms a compound ‘Y’ which on addition of one molecule of hydrogen in the presence of nickel forms a compound ‘Z’. One molecule of compound ‘Z’ on combustion forms two molecules of CO2 and three molecules of H2O. Identify giving reasons the compounds ‘X’, ‘Y’ and ‘Z’. Write the chemical equations for all the chemical reactions involved. [5]
Answer: Organic compound ‘X’ is ethanol, (i.e., CH3 – CH2OH).
When ethanol is heated with conc. H2SO4 it forms ‘Y’ which is ethane (CH2 =CH2).
Question: 20. What is pollination? How does it occur in plants? How does pollination lead to fertilisation? Explain. [5]
Answer: The transfer of pollen grains from anthers (male sexual parts) of a flower to the stigma part of the pistil (female sexual part) is known as pollination. Pollination is done by insects, birds, wind and water.
Pollination can occur in two ways:
(i) Self pollination: When the pollen grains from the anther of a flower are transferred to the stigma of the same flower or another flower on the same plant, it is called self pollination.
(ii) Cross pollination: When the pollen grans from the anther of a flower on one plant are transferred to the stigma of a flower on another similar plant, it is called cross pollination.
When a pollen grain falls on the stigma of the carpel, it grows a pollen tube downwards through the style towards the female gamete in the ovary. A male gamete moves down the tube. When the pollen tube enters the ovule, its tip bursts open and male gamete comes out the pollen tube and combines with the nucleus of the female gamete and forms zygote. This process is known as fertilization.
Question: 21.
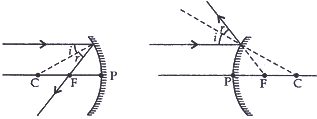
- To construct a ray diagram we use two light rays which are so chosen it is easy to know their directions after reflection. Use these two rays to locate the image of an object placed between infinity and the center of curvature of a concave mirror.
- Draw a ray diagram to show the formation of image of an object placed between the pole and principal focus of a concave mirror. How will the nature and size of the image formed change, if the mirror is replaced by a converging lens of same focal length? [5]
Answer: (a) The rays for obtaining image from the mirror after reflection are so chosen that it is easy to know their directions after reflection from the mirror.
(i) A ray parallel to the principal axis, after reflection passes through the focus in convex mirror or appears to pass through the focus in convex mirror.
(ii) A ray passing through the principal focus of a concave mirror or directed towards the principal focus in convex mirror after reflection will emerge parallel to the principal axis.
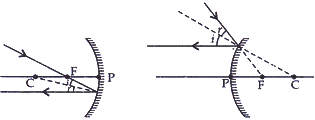
(iii) A ray passing through the center of curvature of the concave mirror or directed in the direction of the centre of curvature of the convex mirror is reflected back along the same path after reflection.
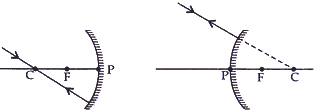
(1) When the object is placed between infinity and the center of curvature of a concave mirror.
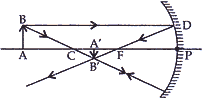
(b). (i) When the object is placed between the pole and principal focus of a concave mirror.
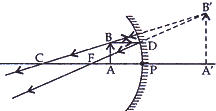
(ii) If the object is placed between the ‘O’ and principal focus of a converging lens (convex lens).
Or
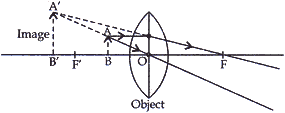
- A student cannot see clearly a chart hanging on a wall placed at a distance of 3 m from his eyes. Name the defect of vision he is suffering from. Draw a ray diagram to illustrate this defect. List its two possible causes.
- Draw a ray diagram to show how this defect may be corrected using a lens of appropriate focal length.
- What do you mean by the ‘Power of Accommodation’ of human eye?
Answer: (a) The student is suffering from Myopia. It is the defect of the eye vision due to which a person can see the near objects clearly, but he cannot see the far objects clearly.
Causes of myopia. Myopia is caused
– due to the elongation of the eye ball.
– due to decrease in the focal length of the eye lens. They lens becomes more convergent.
Myopia can be corrected by using a concave lens of suitable focal length in the spectacles of such a person.
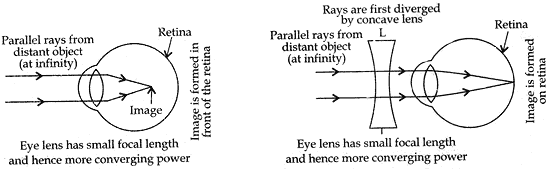
(c) Eye lens can change its focal length according to the distance of the object from the eye, with the help of its ciliary muscles. This phenomenon is known as ‘Power of Accommodation’ of the eye.
Section B
Question: 22.
- What is least count of voltmeter?
- In a voltmeter there are 20 divisions between the ‘0’ mark and 0.5 V mark. Calculate its least count. [2]
Answer: (a) The minimum potential difference measured by a voltmeter between the two given terminals is called the least count of that voltmeter.
(b) Two given marks of the voltmeters = 0 and 0.5 V
Potential difference = 0.5 – 0 = 0.5V
No. of divisions between these two marks = 20
Least count = 0.5/20 = 5/20×20 = 1/40 = 0.025 volts
Question: 23.
- How is pH paper used to find the pH of a solution?
- The pH value of water is 7. What will e the pH value of (i) aqueous solution of sodium hydroxide and (ii) dil HCl. [2]
Answer:
- With the help of a dropper one drop of the solution is placed on the strip of the pH paper. The colour developed on the pH paper is compared with the colour and the corresponding pH value given on the chart of the pH paper.
- (i) aqueous solution of sodium hydroxide is alkaline so its pH will be more than 7.
(ii) dil HCl is acidic so its pH value will be less than 7.
Question: 24. What are the precaution taken to prepare a temporary mount of a leaf peel to show its stomata? [2]
Answer: Precautions taken to prepare a temporary mount of a leaf peel:
- The epidermal peel should be taken from a freshly plucked leaf.
- Always hold the slide by its edge to avoid making the slide dirty.
- Always use a brush to transfer the peel from petri dish to the slide.
- Curling of peel must be avoided.
- The peel should be mounted in the centre of the slide.
- The peel should not be allowed to dry.
- Always keep the cover slip gently to avoid the entry of air bubbles.
Question: 25. Name two salts each of calcium and magnesium which make the water hard? [2]
Answer: These salts are:
Calcium chloride – CaCl2
Calcium sulphate – CaSO4
Magnesium chloride – MgCl2
Magnesium sulphate – MgSO4
Question: 26. Name the process of asexual reproduction shown by yeast. What type of living being is yeast? What is its commercial importance? [2]
Answer: Yeast reproduces by method of budding. Yeast is a unicellular fungus.
Yeast forms lots of buds in the sugar solution. These buds become young yeast cells which change sugar of the solution to produce ethyl alcohol by fermentation in the distilleries.
Question: 27. List the factors on which the angle of deviation through a prism depend? [2]
Answer: angle of deviation through a prism depends
- on the ∠A called ∠ of prism;
- on the ∠ of incidence;
- on the optical density of the material of the prism.
 Class Notes NCERT Solutions for CBSE Students
Class Notes NCERT Solutions for CBSE Students