10 CBSE Science Paper Design 2013-14 Delhi
(Modified Based on Latest CBSE Question Paper Design)
Subject: Science
Time Allowed: 3 Hours
Maximum Marks: 80 General Instructions:
- The Question paper comprises of two sections, A and B. You are to attempt both the sections.
- All questions compulsory.
- All questions of Section A and all questions of Section B are to be attempted separately.
- Question number 1 to 2 in Section A are one mark questions. These are to be answered in one word or in one sentence.
- Question numbers 3 to 5 in Section A are two marks questions. These are to be answered in about 30 words each.
- Question numbers 6 to 15 in Section A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section A are five marks questions. These are to be answered in about 70 words each.
- Question numbers 22 to 27 in Section B are questions based on practical skills and are two marks question.
Section A
Question: 1. Write any method to induce current in a coil. [1]
Answer: A current is induced in a coil when it is moved (or rotated) relative to a fixed magnet.
Question: 2. Mention any one reason due to which most of the thermal plants are set up near coal or oil fields. [1]
Answer: It is easier to transmit electricity over long distances than to carry coal, oil or gas over the same distance, therefore many thermal power plants are established near coal or oil fields.
Question: 3. State the number of water molecules present in crystals of washing soda and plaster of Paris. What are these water molecules called as? [2]
Answer:
- Washing soda crystals contain 10 molecules of water (Na2CO3. 10H2O).
- Plaster of Paris crystals contain ½ molecule of water (CaSO4. ½ H2O or 2CaSO4. H2O).
- These molecules of water are called water of crystals.
Question: Three resistors of 10 Ω, 15 Ω are connected in parallel. Find their equivalent resistance. [2]
Sol. R1 = 10 Ω R2 = 15 Ω R3 = 5 Ω
R1, R2 and R3 are connected in parallel then equivalent resistance (R) is given by
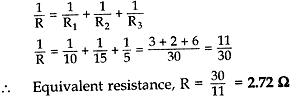
Question: 5. Why is Government of India imposing a ban on the use of polythene bags? Suggest two alternatives to these bags and explain how this ban is likely to improve the environment. [2]
Answer: Plastic bags are non-biodegradable which can not be broken down into non-poisonous substances in nature.
Plastic bags are the main cause of sewer blockages as plastic bags are not decomposed by micro-organisms.
Plastic bags if eaten by stray animals like (like cows) can block their alimentary canal.
Two alternatives:
- We should take own jute or cloth bags while going for purchasing articles from the market.
- Disposal paper cups should be used for serving tea in trains instead of plastic cups.
- Shopkeepers can use paper bags instead of plastic bags.
Question: 6. (i) Calculate the current through a lamb of 25 W operating at 250 V.
(ii) Why elements of electrical hating devices are made up of alloys? [3]
Answer:
(i) Power of the lamp, P = 25 W
Potential difference, V = 250 V Current, I = ?
Formula: P = V × I ∴ 25 = 250 × I ∴ Current, I = 25/250 = 1/10 = 0.1 A
(ii) The heating elements of electrical heating appliances are made up of nichrome alloy because:
(1) nichrome has very light resistivity due to which it produces a lot of heat on passing current.
(2) nichrome does not undergo oxidation easily even at high temperature, it can be kept red hot without burning.
Question: 7. Define the term ‘metallurgy’, and fill appropriated terms in the blanks: [3]The four major steps involved in metallurgy are:
(i) ………… Of ore.
(ii) Conversion of ore into its ………… .
(iii) Reduction of oxides of ores into ……….. .
(iv) ……….. of metal.
Answer: The various processes involved in the extraction of metals from their ores and their refining are known as metallurgy.
The four major steps involved in metallurgy are:
(i) Concentration of ore.
(ii) Conversion of ore into its metal oxide.
(iii) Reduction of oxides of ores into metal.
(iv) Refining of impure metal.
Question: 8. Find the resistance between points A and B in the circuit diagram given below: [3]
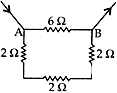
Solution:
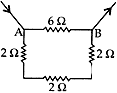
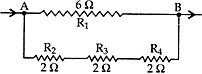
R1 = 6 Ω
R2 R3 R4 are in series and have resultant resistance R’
R’ = R2+ R3+ R4 = 2 + 2 + 2 = 6 Ω
R’ is in parallel combination with R1
∴ Resultant resistance of the circuit (R)
⇒ 1/R = 1/R1 + 1/R1 = 1/6 + 1/6 = 2/6 = 1/3
∴ Resistance, R = 3 Ω
Or
(a) Describe an activity to show with the help of a compass that magnetic field is strongest near poles of a bar magnet.
(b) Mention the direction of magnetic field lines (i) inside a bar magnet and (ii) Outside a bar magnet.
Answer: (a) A bar magnet is placed on a paper and its boundary is marked with a pencil. A magnetic compass is brought near the N-pole of the bar magnet. It is observed that N-pole of magnet repels the N-pole of compass needle due to which the tip of the compass needle moves away from the N-pole.
Thus a magnetic field pattern is obtained around a bar magnet. Each magnetic field line is directed from the north pole of a magnet to its south pole. The field lines are closest together at the two poles of the bar magnet. The strength of magnetic field is indicated by the degree of closeness of the field lines. So the magnetic field is the strongest near the poles.
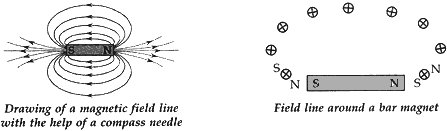
(b) (i) The direction of magnetic field lines inside a bar magnet is from its south pole to its north pole.
(ii) The direction of magnetic field lines outside a bar magnet is from its north pole to its south pole.
Question: 9. Give reasons for the following:
(1) Arteries are thick walled.
(2) Blood goes only once through the heart in fishes.
(3) Plants have low energy needs. [3]
Answer:
- Arteries are thick walled because these vessels carry blood from the heart to all the parts of the body blood emerges from the heart under high pressure.
- A fish has a two chambered heart and has gills to oxygenate blood. In a fish, the heart pumps deoxygenated blood from the gills is supplied to the body parts of the fish where oxygen is utilised and carbon dioxide enters into it, making it deoxygenated. This deoxygenated blood returns to the heart to be pumped into gills again. Thus blood passes through the heart of fish only once in one complete cycle.
- Plants are fixed at a place and do not show any locomotion. Plants are less active. Their cells do not need to be supplied with materials so quickly. Therefore plants have low energy needs and respire at a slow rate.
Question: 10. Write the basic principle involved for producing electric energy in a nuclear power plant. Write two problems faced by the people living near such nuclear power plants. [3]
Answer: Principle. Controlled ‘Chain fission’ reaction is carried in the reactor of the nuclear power plant to produce heat energy. This heat energy is then used to produce steam.
The heat energy of the steam is then changed to kinetic energy to rotate the turbine blades and then electric energy is generated by the generators of the power plant.
Problems faced by the people living near nuclear power plants. People living near the nuclear power plants may suffer from many diseases in case of leakage of harmful radiations from the plant.
If nuclear wastes of the plants are not disposed off properly, they may cause birth defects. skin cancers, miscarriages and genetic diseases in the people living around the area.
Question: 11. State the meaning of functional group in a carbon compound. Write the functional group present in (i) ethanol and (ii) ethanoic acid and also draw their structures. [3]
Answer: An ‘atom’ or ‘a group of atoms’ which makes a carbon compound reactive and decides its properties (or functions) is called a functional group.
(i) Ethanol → Alcohol group (— OH)
(ii) Ethanoic acid → carboxylic acid group (— COOH)
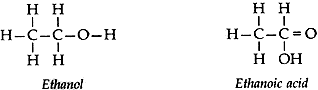
Question: 12. Based on the group valency of elements state the formula for the following giving justification for each: [3](i) Oxides of 1st group elements,
(ii) Helides of the elements of group 13, and
(iii) Compounds formed when an element of group 2 combines with an element of group 16.
Solution:
(i) Oxides of 1st group element (Let it be A):
1st group element: A Valency: +1
Oxide: O Valency: -2
∴ Chemical Formula:
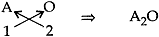
(ii) Halides of the elements of group 13 (Let it be M):
Let element of group 13: M Valency: +3
Halide: X Valency: -1
∴ Chemical Formula:
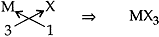
(iii) Compounds of element of group 2 combines with an element of group 16 (Y):
Element of group 2: X Valency: +2
Element of group 16: Y Valency: -2
∴ Chemical Formula:
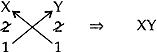
Or
(1). Define the following terms:
(i) Valency; (ii) Atomic size
(2). How do the valency and the atomic size of the elements vary while going from left to right along a period in the modern table?
Answer:
- (i) Valency: The combining capacity of an element is called its valency.
(a) The valency of an element s determined by the number of valence electrons present in the atom of the element.
(b) The number of electrons lost or gained or shared by one atom of an element to achieve the nearest inert gas configuration gives the valency of the element.
(ii) Atomic size: The atomic size of an atom is the distance between the center of the nucleus and outermost electron shell of an isolated atom. - (i) On moving from left to right along a period in the modern periodic table, the valency of elements increases from 1 to 4 and then decreases to zero.
(ii) On moving from left to right in a period of the period table, the size of the atoms decreases.
Question: 13. (1) Give the evidence that the bird have evolved from reptiles.
(2) Inserts, octopus, planaria and vertebrates possess eyes. Can we group these animals together on the basis of eyes that they possess? Justify your answer giving reason. [3]
Answer:
- A fossil bird called Archaeopteryx looks like a bird but it has many features which are found in reptiles. Archaeopteryx has feathered wings like birds but teeth and tail like those of reptiles. Therefore Archaeopteryx is a connecting link between the reptiles and birds and hence suggests that thee birds have evolved from the reptiles.
- Inserts, octopus, planaria and vertebrates posses eyes but these animals can not be put together on the basis of eyes because the structure of eyes in each of there organisms is different which suggests their separate evolutionary origins. Thus all these animals are put in different groups.
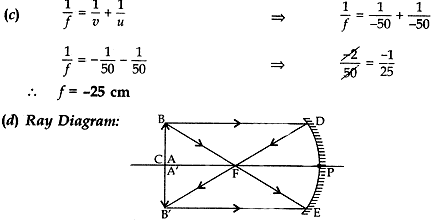
Question: 14. A spherical mirror produces an image of magnification -1 on a screen placed at a distance of 50 cm from the mirror.
(a) Write the type of mirror.
(b) Find the distance of the Image from the object.
(c) What is the focal length of the mirror?|
(d) Draw the ray diagram to show the image formation in this case.
Answer: If magnification, m = -1
v = 50 cm
If the magnification has minus sign, then the image is real and inverted.
∴ v = -50 (for real image)
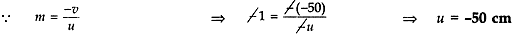
(a) Since image is formed on the screen therefore the mirror formed real image which is formed by concave mirror only.
(b) Image distance = 50 cm in front of the mirror.
Question: 15. Explain giving reason why the sky appears blue to an observe from the surface of the earth? What will the colour of the sky be for an astronaut staying in the international space station orbiting the earth? Justify your answer giving reason. [3]
Answer: Sky appears blue: The molecules of air and other fine particles in the atmosphere have a size smaller than the wavelength of visible light. So these particles scatter more effectively the light rays of shorter wavelength at the blue end than light of longer wavelengths at the red end. When the scattered blue light enters our eyes, it gives us the feeling of a blue sky.
Color of sky to an astronaut: There is no atmosphere containing air in space to scatter sunlight. As there is no scattering of light in space, the scattered light does not reach the eyes and the sky appears dark instead of blue to an astronaut in outer space.
Question: 16. Pure iron is soft and stretches easily when hot
(1). How does this property of iron change when:
(i) small amount of carbon js mixed with it?
(ii) nickle and chromium are mixed with it?
(2). Define an alloy. How is an alloy prepared?
(3). An alloy has low melting point and is therefore used for electrical fuse. Name the alloy and write its constituents. [5]
Answer:
- (i) When small amount of carbon is mixed with iron, it becomes an alloy that is called steel. The carbon present in pig iron or cast iron makes it brittle and therefor, it cannot be used for structural purposes.
(ii) When iron is mixed with chromium and nickel, then stainless steel is obtained. Stainless steel does not rust at all.
Stainless steel is used for making utensils, knives, scissors and surgical instruments etc. - Alloy: An alloy is a homogeneous mixture of two or more metals or a metal and a non-mental. An alloy is generally prepared by heating the mixture of the metals (or metals and non-metals) to the molten state and then cooling the mixture to solidify at room temperature.
- A fuse wire is made up of tin and copper or tin and lead (composition: 63% tin and 37% lead).
Question: 17. (a) Differentiate between AC and DC. Write any two points of difference.
(b) A person operates a microwave oven of 2kW power rating in a domestic circuit of 220 V and current rating 5A. What result is expected? Explain with reason.
(c) Write the frequency of AC and DC. [5]
Answer: (a)
| AC | DC |
| (i) If the current reverses its direction after equal intervals of time, it is called alternating current (AC). | (i) If the current flows in one direction only, it is called direct current (DC). |
| (ii) Some sources of alternating current are power house, generators, car alternators and bicycle dynamos. | (ii) Sources of direct current are dry cell, dry cell battery, car battery. |
(b) Power of the oven, P = 2 kW = 2000 W
Potential difference, V = 220 volt
Current, I = 5 A
∴ Result will be:
P = V × I = 220 × 5 = 1100 volt A = 1100 W
This shows that microwave requires more electrical energy than the energy provided by the circuit. The microwave will not work and fuse wire will break due to overloading.
(c) The AC produced in India has a frequency of 50 Hz, it means the current reverses its direction 100 times in a second.
DC always flows in one direction, it means polarity of direction current is fixed and it has no frequency.
Question: 18. Draw the human respiratory system and label the following parts: [5](a) Trachea (b) Alveoli (c) Respiratory bronchioles (d) Larynx
Describe in brief the role of lungs in the exchange of gases.
Answer: Human respiratory system:
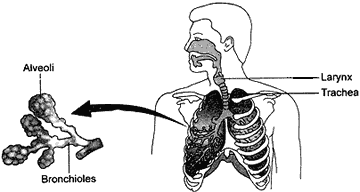
Role of lungs:
- When we breather in air, the diaphragm contracts which result in the increase of chest cavity. Due to this expansion of chest cavity, the air pressure in the lungs decreases. Thus, air from outside rushes into lungs through nostrils, trachea and bronchi. Thus sacs of lungs get filled with air when we breathe in. The exchange of gases between alveoli and blood takes place by the process of diffusion.
- Now the air present in air sacs of the lungs is rich in CO2. When we breathe out air, the diaphragm relaxes which result in the decrease of chest cavity. This contraction pushes the air from the lungs into the trachea, nostrils and then out of the body into air. Breathing in of air is called inhalation and breathing out of air is called exhalation.
Question: 19. State the reason why carbon can neither form C4+ cations nor C4- anions, but forms covalent compounds. Also state reasons to explain why covalent compounds: [5](1). are bad conductors of electricity?
(2). have lows melting and boiling points?
Answer: Carbon atoms have 4 electrons in their outermost shell. So needs to gain of lose electrons to attain noble gas configuration.
(i) It could gain four electrons forming C4- anion. But it would be difficult for the nucleus with six protons to hold on to ten electrons.
(ii) It could lose four electrons forming C4+ cation. But it would require a large amount energy to remove four electrons from its outermost shell.
Because of these reasons, carbon shares valence electrons to complete its octet with other atoms to form covalent bonds.
- Covalent compounds are bad conductors of electrons to complete its octet with other atoms to form covalent bonds.
- Covalent compounds have usually low malting and boiling points because they are made up of electronically neutral molecules. So the force of attraction between the molecules of a covalent compound is very weak. Only a small amount of heat energy is required to break these weak molecular forces, due to which covalent compounds have low melting and boiling points.
Question: 20. (1) Give one example each of a unisexual and a bisexual flower.
(2) Mention the changes a flower undergoes after fertilization.
(3) How does the amount of DNA remain constant though each new generation which is a combination of DNA copies of two individuals? [5]
Answer:
- Uni-sexual Flower – Papaya
Bisexual Flower – Hibiscus (China Rose) - After fertilization, the fertilized egg divides several times to form an embryo within the ovule. The ovule develops a tough coat around it and is gradually converted into a seed (containing the baby plant along with food). The ovary of flower develops and becomes a fruit (with seeds inside it). The other parts of flower like sepals, petals, stamens, stigma and style dry up and fall off. Only the ovary is left behind.
- In sexual reproduction, though the genetic material DNA from two gamete, male and female gametes, combine together to form a new cell ‘zygote’ but the amount of DNA in zygote does not get doubled. This is because the gametes are special type of cells which contain only half the amount of DNA as compared to the normal body cells of an organism. So the new cell zygote formed by two gametes cells will have the normal amount of DNA.
Or
(1). Name the respective part of human female reproductive system:
(i) that produces eggs.
(ii) where fusion of egg and sperm takes place, and
(iii) where zygote get implanted.
(2). Describe i brief what happens to the zygote after it gets implanted.
Answer:
- Part of human female reproductive system:
(i) that produces eggs – Overy
(ii) where fusion of egg and sperm takes place – Fallopian tube
(iii) where zygote get implanted – Uterus - After zygote gets implanted, a disc like special tissue develops between the uterus wall and embryo (foetus) which is called placenta. The foetus is connected to the placenta in the mother’s body through the umbilical cord. The exchange of nutrients, oxygen and waste products between the embryo and the mother takes place through the placenta.
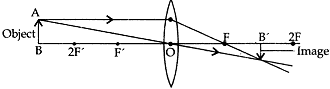
Question: 21. A student wants to project the image of a candle flame on the walls of school laboratory by using a lens:
(a) Which type of lens should he use and why?
(b) At what distance in terms or focal length ‘F’ of the lens should he place the candle flame so as to get (i) a magnified, and (ii) a diminished image respectively on the wall?
(c) Draw ray diagram to show the formation of the image in each case. [5]
Answer: (a) A convex lens should be used. This is because it can produce a real image of the candle lame on the wall as it is a converging lens where refracted rays actually meet.
(b) & (c) (i) A real magnified image is formed when the candle flame is placed between F and 2F from the convex lens on the other side of the wall.
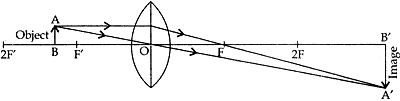
(ii) A real diminished image is formed when the candle flame is flame is placed beyond 2F from the convex lens on the other side of the all.
Section B
Question: 22. The reading of current flowing through a conductor and the potential difference across its two ends are shown in the ammeter and voltmeter given below. What will be the value of resistance in it? [2]
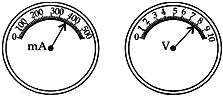
Sol. I = 400 mA = 400/1000 = 0.40 A, V = 8 volts
According to Ohm’s Law = V = IR
⇒ R = V/I
= 8 × 10/4 = 20 Ω
Question: 23. Two resistances R1 = 10 ohm and R2 = 10 ohm are to be connected with 20V battery. Out of which of the following maximum current will flow. Calculate its magnitude. [2]
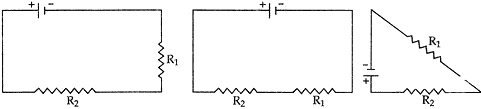
Answer: In all the three cases, the same current will be flowing as in all the three cases, both the resistors are combined in series.
R1 = 10 Ω R2 = 10 Ω
Resultant R = R1 + R2 = 10 + 10 = 20 Ω
V = 20 V
I = V/R = 20/20 = 1A
Question: 24. Match the important chemical given in column (A) with the chemical formula given in Column (B).
| Column (A) | Column (B) |
| (a) Plaster of Paris (b) Gypsum (c) Bleaching powder (d) Slaked lime |
(i) Ca(OH)2 (ii) CaSO4. ½H2O (iii) CaOCl4. 2H2O (iv) CaOCl2 |
Answer:
| Column (A) | Column (B) |
| (a) Plaster of Paris (b) Gypsum (c) Bleaching powder (d) Slaked lime |
(ii) CaSO4. ½H2O (iii) CaOCl4. 2H2O (iv) CaOCl2 (i) Ca(OH)2 |
Question: 25. What do you observe when dil. HCl is added on Zn pieces taken in a test tube? How would you identify the gas evolved? Write one use of this gas. [2]
Answer: Observation: A colourless gas is evolved with brisk bubbles from the test tube.
When a burning match stick is brought near the gas, then the gas burn with a pop sound. The gas evolved is H2 gas.
H2 gas is used as fuel in the space crafts.
Question: 26. What do you observe on seeing the slides showing reproduction in amoeba? What is the name given to this method of reproduction in Amoeba? [2]
Answer: On observing this slide, we see different stages under which the nucleus of amoeba cell is dividing into two parts.
Method of reproduction in amoeba. Binary fission
Question: 27. For finding the focal length of a concave mirror, where do we keep the object? What is the position of image formed? On which structure we get the image? What is the nature of the image formed?
Answer: A large sized object placed at a far off (Infinity) place, seen from the window of the lab, is taken as object.
The image is formed at F point of the mirror. This image is formed on the screen.
The distance between the mirror and the screen is measured; which is equal to focal length or the mirror. The image formed is real and inverted.
 Class Notes NCERT Solutions for CBSE Students
Class Notes NCERT Solutions for CBSE Students